L'herència genètica dels denissovans pot haver deixat empremta en la nostra salut mental
Un equip de recerca liderat per l'Institut de Biologia Evolutiva (IBE) i la Universitat Pompeu Fabra (UPF) ha identificat la contribució genètica més estesa dels denissovans fins avui.
L'estudi assenyala que la variant genètica observada, que afecta la regulació del zinc, va poder representar un avantatge evolutiu en l'adaptació al fred dels nostres ancestres.
El treball, que publica Plos Genetics, també revela que aquesta adaptació genètica va poder predisposar als humans moderns a sofrir trastorns neuropsiquiàtrics.
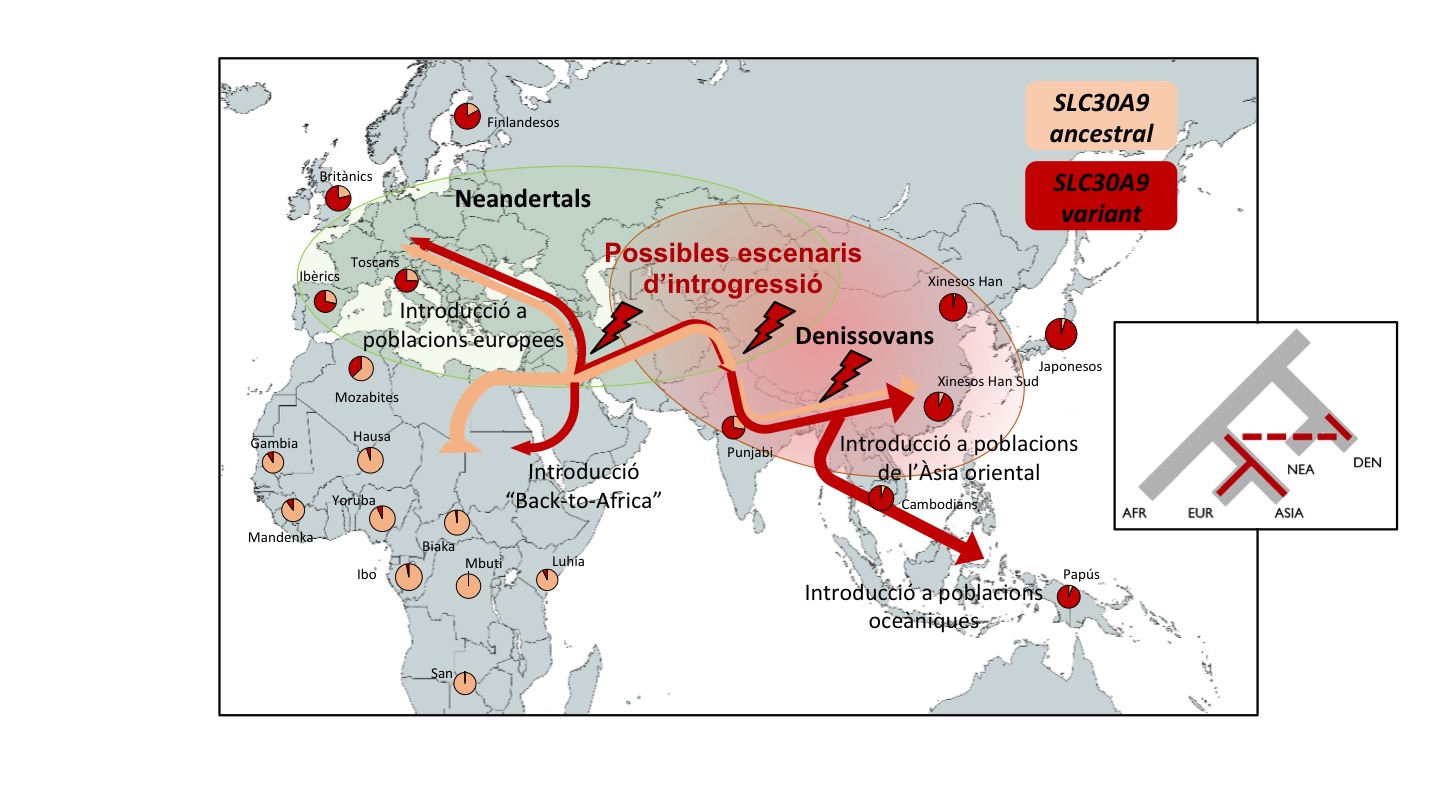
Els humans moderns van sortir d'Àfrica en l'esdeveniment conegut com a "Out-of-Africa" fa 60.000 anys. Un cop a Àsia, van coincidir amb els denissovans i, d'aquesta trobada, possiblement van sorgir enfrontaments i col·laboracions, però també diversos encreuaments. De fet, encara avui dia els humans moderns conservem en el nostre genoma variants genètiques d'origen denissovà, que són testimoniatge d'aquells primers acostaments.
Ara, un equip liderat per l'Institut de Biologia Evolutiva (IBE), un centre mixt del Consell Superior d'Investigacions Científiques (CSIC) i la Universitat Pompeu Fabra (UPF), i pel Departament de Medicina i Ciències de la Vida (MELIS) de la UPF, ha identificat una de les petjades de l'herència genètica dels extints denissovans més estesa en els humans moderns. Els equips d'Elena Bosch, investigadora principal de l'IBE, i de Rubén Vicente, investigador principal del MELIS-UPF, han descobert que aquesta adaptació genètica va poder ajudar les poblacions ancestrals de sapiens a adaptar-se al fred.
La variant observada, implicada en la regulació del zinc i amb un paper en el metabolisme cel·lular, podria també haver predisposat als humans moderns a patir trastorns psiquiàtrics com la depressió o l'esquizofrènia.
La variació genètica en la regulació del zinc va poder suposar un avantatge evolutiu
Com l'adaptació ha donat forma a la diversitat genètica actual en les poblacions humanes és una qüestió de gran interès en genètica evolutiva. Arran d'aquesta pregunta, l'equip d'Elena Bosch va identificar una variant adaptativa entre poblacions humanes actuals en una regió del nostre genoma que presenta gran similitud al genoma d'una població ancestral i extinta: els denissovans.
"A través de l'anàlisi genòmica, identifiquem que la variant genètica observada provenia del nostre encreuament amb humans arcaics en el passat, possiblement els denissovans", comenta Ana Roca-Umbert, co-primera coautora de l'estudi. L'equip ha pogut descartar que l'herència fos neandertal, atès que aquestes poblacions no presenten aquesta mutació.
"Pel que sembla, el canvi va resultar beneficiós i va suposar un avantatge selectiu per als humans. Com a conseqüència, aquesta variació al gen SLC30A9 va ser seleccionada i ha arribat fins a les poblacions actuals", afegeix Jorge Garcia-Calleja, co-primer coautor de l'estudi.
El Laboratori de Genètica Evolutiva de Poblacions, que dirigeix Bosch, va voler esbrinar quins canvis provoca a escala cel·lular aquesta variació genètica d'origen denissovà. "Descobrim que aquesta mutació segurament tenia implicacions en el transport del zinc dins de la cèl·lula, i llavors contactem amb l'equip de Vicente", relata Elena Bosch, investigadora principal de l'IBE i corresponsable de l'estudi.
La regulació del zinc: clau per a l'adaptació al fred
"Elena em va contactar perquè el seu equip havia observat un canvi en un aminoàcid en un transportador de zinc, que era molt diferent entre les poblacions de l'Àfrica i l'Àsia actuals. A partir d'allí, comencem a fer-nos preguntes i a buscar respostes", comenta Rubén Vicente. El seu equip, en el grup de Biofísica del Sistema immunitari en el Laboratori de Fisiologia Molecular, va assumir el repte tècnic d'estudiar el moviment del zinc intracel·lular.
El zinc, oligoelement essencial per a la salut humana, és un missatger important que trasllada tanta informació de l'exterior cap a dins de les cèl·lules com entre diferents compartiments cel·lulars. La seva manca provoca alteracions de creixement, neurològiques i immunitàries, encara que "la seva regulació encara està poc estudiada per la falta d'eines moleculars per a seguir el flux del zinc".
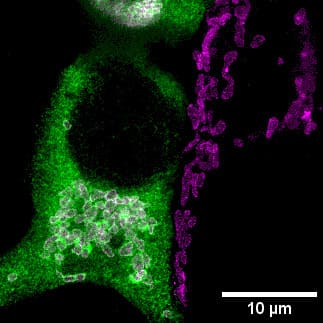
Imatge 1: Demostració de què part del transportador es troba en els mitocondris. Imatge de superresolució amb microscòpia STED de cèl·lules HEK293 transfectades amb el transportador de zinc ZnT9 (en verd). En magenta la proteïna mitocondrial TOM 20 i en blanc la localització a mitocondri. Crèdit: Rubén Vicente.
El laboratori de Vicente va identificar que la variant observada provoca un nou equilibri del zinc dins de la cèl·lula, promovent un canvi al metabolisme. En alterar el reticle endoplasmàtic i els mitocondris de les cèl·lules, aquesta variació provoca un possible avantatge metabòlic per a fer front a un clima hostil. "El fenotip observat ens fa pensar en una possible adaptació al fred", comenta Vicente.
L'herència genètica denissovana podria afectar la salut mental de les poblacions europees i asiàtiques
El transport del zinc també està implicat en l'excitabilitat del sistema nerviós, i té un paper en l'equilibri i la salut mental de les persones.
L'equip apunta al fet que la variant trobada en aquest transportador de zinc, que s'expressa en tots els teixits del cos, s'associa a una major predisposició a sofrir algunes malalties psiquiàtriques. Entre elles, es troben l'anorèxia nerviosa, el trastorn d'hiperactivitat, el trastorn de l'espectre autista, el trastorn bipolar, la depressió, el trastorn obsessiu compulsiu (TOC) i l'esquizofrènia.
"En el futur, estendre aquest estudi a models animals podria posar llum sobre aquesta predisposició a patir malalties mentals", apunta Vicente.
La variant genètica ha deixat una empremta global, excepte a Àfrica
Encara que la variant es fixés a Àsia arran dels encreuaments entre denissovans i sàpiens, aquesta s'ha estès també a Europa i poblacions natives americanes. De fet, es troba a poblacions de tot el planeta, encara que, en el cas de les poblacions africanes, és molt menys freqüent.
L'equip apunta que, probablement, es tracta de l'adaptació genètica denissovana de major abast geogràfic descoberta fins ara. "Per exemple, una variant en el gen EPAS1 heretada dels denissovans permet l'adaptació a la vida en altitud, però es troba només a la població tibetana. No obstant això, en el nostre cas l'impacte s'estén en totes les poblacions fora d'Àfrica", conclou Bosch.
Estudi referenciat: Ana Roca-Umbert , Jorge Garcia-Calleja , Marina Vogel-González, Alejandro Fierro-Villegas, Gerard Ill-Raga, Víctor Herrera-Fernández, Anja Bosnjak, Gerard Muntané, Esteban Gutiérrez, Felix Campelo, Rubén Vicente, Elena Bosch. Human genetic adaptation related to cellular zinc homeostasis. Plos Genetics; DOI: https://doi.org/10.1371/journal.pgen.1010950
